In Situ pH Control
On this page:
- Schematic
- Introduction
- Other Technology Names
- Description
- Development Status
- Applicability
- Cost
- Duration
- Implementability Considerations
- Resources
Schematic
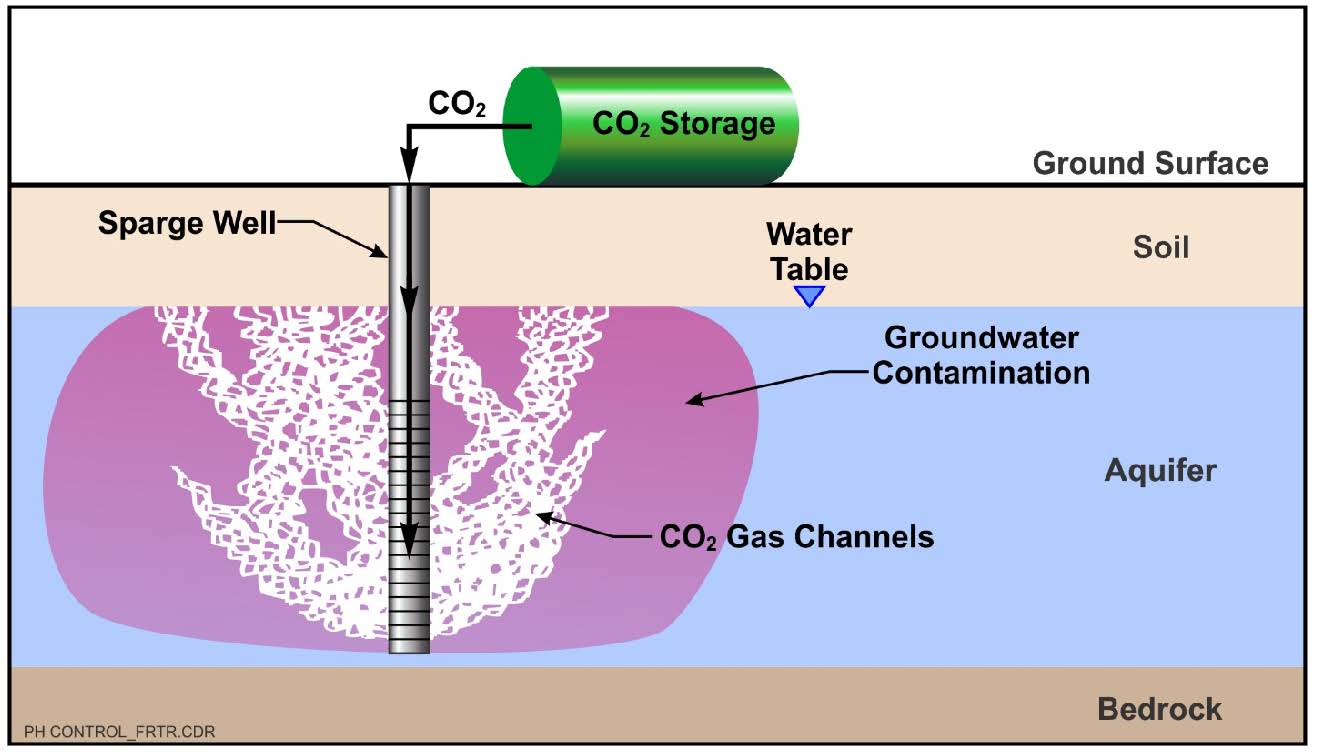
This information may be reproduced without restriction as long as the source attribution is included.
In Situ pH Control with Carbon Dioxide Sparging
Introduction
pH control is an in situ technology used to neutralize soil and groundwater having high or low pH. It can be used as a stand-alone technology at sites where the aquifer pH has been impacted due to releases of acidic or caustic reagents or can be used in conjunction with other remedies, such as reductive dechlorination, that can alter the pH of the aquifer during application. Carbon dioxide (CO2) sparging is one type of application that can be used to neutralize contaminant plumes with high (basic) and low (acidic) pH. CO2 introduced into the aquifer reacts with groundwater to produce carbonic acid, which lowers the pH of the aquifer. Other types of pH control include the introduction of water amended with acids, bases, or buffers through injection wells or points.
Other Technology Names
Neutralization
Description
In situ pH control may be required at a site for one or more of the following reasons:
- Groundwater pH is very low or very high due to the historical release and/or disposal of various reagents and must be treated based on local regulatory requirements.
- High concentrations of metals are present in groundwater, which can be precipitated by manipulating pH (and oxidation reduction potential).
- The application of a remedy such as reductive dechlorination or in situ chemical oxidation (ISCO) changes the pH to a value that is detrimental to the effectiveness of the technology or does not comply with regulatory requirements.
- Fouling (scaling) of wells, the formation, equipment, and fluid conveyance lines is known or expected to occur during application of pump and treat systems and other remedies that require the extraction, treatment, and recirculation of groundwater.
These high or low pH conditions can be treated through the addition of buffers, carbon dioxide sparging, and addition of acids and bases, which are described below.
Buffers
Buffers are often required to maintain a neutral pH during remediation. All aquifers have a certain amount of buffering capacity, which varies based on site-specific geochemistry. The most common naturally-occurring buffer is dissolved CO2 and the reversible reactions that it undergoes to form carbonate and bicarbonate ions, which react with the hydrogen ions and neutralize high or low pH water. Because the reaction is reversible, high pH water will cause carbonate and bicarbonate ions to disassociate to produce hydrogen ions, which serve to lower (neutralize) the high pH water, while low pH water will cause bicarbonate and carbonate ions to protonate (i.e., consume a hydrogen ion), which serves to increase the pH.
Naturally-occurring minerals also will contribute to the buffering capacity of an aquifer. In particular, calcium carbonate (i.e., the mineral calcite) will dissolve and release carbonate ions in the presence of acidic groundwater, which similarly react with the hydrogen ions. Conversely, alkaline water will disassociate bicarbonate and carbonate ions to produce hydrogen ion and lower the pH of the aquifer. In addition to these reactions, there are other generally less pronounced reactions involving phosphates, silicates, borates and various clay minerals that also contribute to the buffering capacity of an aquifer (SERDP-ESTCP, 2019).
There are instances when the natural buffering capacity is not sufficient to overcome pH changes created by the application of a remedial technology. For instance, during reductive dechlorination, the hydrogen ion is produced, from biodegradation of the COC and electron donor, which can lower the pH of the aquifer to values that inhibit microbial activity, and therefore must be neutralized.
Several commercially available amendments are available for in situ application to address high or low pH groundwater. Properties of these amendments vary and should be carefully considered based on site-specific conditions and remedial goals. For instance, sodium bicarbonate, readily available from various suppliers, is highly soluble in water and is inexpensive. Because of its high solubility, it is easily dispersed from the treatment area, and may require frequent replenishment. It may not be suitable at sites having high groundwater velocities. AquabupH, another commercially available but proprietary amendment, consists of an alkaline buffer suspended in an emulsified oil. It is more recalcitrant than sodium bicarbonate and therefore is better suited at sites that have a higher rate of influx of groundwater into the treatment area. Another proprietary formulation is Neutral Zone, consisting of a colloidal suspension of calcium carbonate, which is highly recalcitrant and can be used when there is a high influx of groundwater and/or when it is not practical to frequently replenish the amendment in the aquifer. Other minerals and reagents used to buffer and raise the pH include soda ash, hydrated lime, phosphates, and silicate minerals among others. Recycled concrete also has been used in permeable reactive barriers (PRBs) to treat acidic water resulting from pyrite oxidation of acid sulfate soils (Indraratna et al., 2010).
Amendments generally are added in liquid form. Solids, such as silicates and carbonates, can be introduced as a colloidal suspension. Click here for additional guidance to introduce and distribute amendments into the aquifer.
Groundwater acidity should be measured to determine the amount of buffer needed to neutralize the groundwater at a site. Acidity may be measured by Standard Method 2310, in which a water sample is titrated to a specified end point with a strong base (APHA et al., 2017). The quantity of amendments can then be determined based on the result. In cases where the remediation itself is expected to change the pH of the aquifer, additional calculations must be performed. The publicly available software BUCHLORIC (Robinson et al., 2009) can be used to estimate dosing requirements for sodium bicarbonate. In addition, amendment vendors will perform calculations based on the remedial application and site-specific properties and/or will provide software for calculating dosing requirements of their amendments.
Carbon Dioxide Sparging
CO2 sparging is a remedial technology used to introduce CO2 into the aquifer, which then buffers the aquifer as described previously. It has been effectively applied to neutralize groundwater having an average pH of greater than 11 prior to the remedial action (Mutch, 2016). It is used to neutralize either high or low pH and/or to remediate high levels of dissolved metals in groundwater, and oftentimes is used in conjunction with other technologies that impact aquifer pH to provide additional buffering capacity.
CO2 sparging is implemented in the same manner as air sparging, the primary difference being that CO2 is introduced into the aquifer in lieu of air. The injected CO2 migrates through the soil in discrete channels (not as bubbles). Some removal of volatile organic compounds (VOCs) from the groundwater may occur if present and a vapor recovery system may be required to prevent fugitive emissions from occurring during application at some sites.
CO2 is easily transported to a site by refrigerated tanker truck and supplied in liquid form. Heated vaporizers are used to convert the liquid to a gas, which is transferred through a distribution manifold into injection points or wells. Pressure regulators, flow meters, and auxiliary heaters are used to control injection pressure, flow rate and temperature to achieve the required design mass loading at each location. Similar to air sparging, pulsed flow can be applied to optimize introduction and distribution of the CO2 into the aquifer.
Process monitoring should be performed throughout the application. Groundwater levels should be measured to evaluate the degree that groundwater mounding is occurring. Alkalinity, total dissolved solids, total metals, and groundwater quality parameters (dissolved oxygen, oxidation reduction potential, conductivity, temperature and pH) should be assessed to evaluate distribution and understand impacts the application has on the aquifer. Changes in CO2 concentrations in groundwater also can be monitored to assess distribution.
Acids and Bases
Strong or weak acids and bases may be added to the aquifer to rapidly change aquifer pH and/or treat groundwater or process water to prevent fouling or scaling of process equipment, conveyance piping and wells. Acids and bases also are sometimes used to adjust aquifer pH to facilitate application of another remedial technology. For instance, citric acid has been used to reduce aquifer pH to promote ISCO using Fenton's reagent (the catalytic reaction of hydrogen peroxide and iron). Acids and bases also can be used during soil flushing. Acidic solution can be used to remove metals and some organic contaminants, while bases can be used to remove phenols and some metals (GRTAC, 1997).
Treatment of acidic (low pH) groundwater can be performed by adding a variety of caustic materials to the aquifer. In addition to various buffers as described above, common bases include caustic soda (NaOH), caustic potash (KOH), soda ash (Na2CO3), sodium bicarbonate (NaHCO3), hydrated lime (Ca(OH)2) and milk of magnesia (Mg(OH)2) (SERDP and ESTCP, 2019). These salts are commonly dissolved into water and introduced in liquid form into the aquifer. The choice of base will depend on site conditions and project objectives.
Low pH groundwater is more commonly encountered at sites, but high pH groundwater can be present in aquifers beneath steel mills, slag dumps, chlor-alkali plants, or other sites where high pH material has been released or buried. Buffering to reduce aquifer pH (e.g., CO2 sparging) is preferred, but weak acids can be added to reduce pH if needed. Care must be taken not to overdose and to achieve adequate distribution of the acid to prevent areas of low pH groundwater to persist, which can have adverse impacts on the aquifer (e.g., dissolution of naturally occurring metals). Commonly used acids include citric, nitric, and hydrochloric, among others. Solutions are easily prepared aboveground and are injected and further diluted in the subsurface.
The appropriate dosage of acids and bases must be determined prior to application. Titration tests using site-specific soil and groundwater may be performed to determine acid demand and estimate the quantity of acid or base required to achieve a target pH at a given alkalinity. Pilot tests also may be performed to determine the type and dosage of amendment needed and identify any site-specific challenges associated with the application.
Development and Implementation Status
The following checklist provides a summary of the development and implementation status of pH control:
☐ At the laboratory/bench scale and shows promise
☐ In pilot studies
☒ At full scale
☐ To remediate an entire site (source and plume) focused on plume
☒ To remediate a source only
☒ As part of a technology train
☐ As the final remedy at multiple sites
☐ To successfully attain cleanup goals in multiple sites
pH control is available through the following vendors:
☒ Commercially available nationwide
☐ Commercially available through limited vendors because of licensing or specialized equipment
☐ Research organizations and academia
Applicability
|
Contaminant Class Applicability Rating for Cometabolic Bioremediation (Rating codes: Demonstrated Effectiveness, ◐ Limited Effectiveness, No Demonstrated Effectiveness, Level of Effectiveness dependent upon specific contaminant and its application/design, I/D Insufficient Data, N/A Not Applicable) |
||||||||
|---|---|---|---|---|---|---|---|---|
Nonhalogenated VOC |
Halogenated VOC |
Nonhalogenated SVOC |
Halogenated SVOC |
Fuels |
Inorganics |
Radionuclides |
Munitions |
Emerging Contaminants |
| ● | ● | ● | ● | ○ | ● | ● | ● | I/D |
pH control is used to address high or low pH groundwater and contaminants of concern, such as metals and radionuclides, which exhibit different solubilities at different pH values. Used in conjunction with other technologies such as ISCO, reductive dechlorination, and soil washing, it can facilitate removal of a wide range of contaminants such as halogenated and non-halogenated VOCs and SVOCs, radionuclides, and munitions constituents. pH control generally is not used to facilitate removal of fuels.
Cost
pH control is an aggressive technology that is used to modify the pH of the aquifer either as a standalone technology or in support of other remedial technologies. Major cost drivers include:
Upfront Costs
- Physical and chemical characteristics of the contaminated soil including buffering capacity, grain size and types and concentrations of COCs, which will impact the type of treatment required to achieve project goals.
- Target treatment volume.
- Application design and treatment requirements. For instance, CO2 sparge systems will be more costly than a pH control system designed to mitigate fouling in wells, conveyance lines and equipment. CO2 sparge systems that require vapor treatment will be more costly than those that don't.
Operation and Maintenance Costs
- Reagent dosage requirements.
- Frequency of reapplication, and volume and type of amendments.
- The duration of each injection event, the type of technology implemented, the method of application and monitoring requirements.
- Vapor collection and treatment during CO2 sparging, if required necessitate additional labor, utilities, sampling and analytical costs.
- Areal extent of contamination. Larger sites necessitate more sparge points, resulting in greater air requirements and energy.
- In the case of CO2 sparge systems, sites at which the depth of contamination beneath the water table is greater will require higher pressures to introduce CO2 and therefore compressor capacity and utility costs are greater.
The list above highlights those cost dependencies specific to pH control and does not consider the dependencies that are general to most in situ remediation technologies. Click here for a general discussion on costing which includes definitions and repetitive costs for remediation technologies. A project-specific cost estimate can be obtained using an integrated cost-estimating application such as RACER® or consulting with a subject matter expert.
Duration
pH control is a short- to medium-term treatment technology expected to require weeks to months to treat the contaminated media. However, treatment duration is largely based on the type of treatment applied, project objectives, and remedial goals. For instance, CO2 sparge systems may require several months of sparging followed by a monitoring period and possibly additional sparging at a later date should rebound of pH (or metals) occur. The duration of pH control performed to support applications such as ISCO, reductive dechlorination, soil flushing, and others is based on the expected duration of those technologies.
Implementability Considerations
The following are key considerations associated with pH control:
- Naturally occurring metals may be mobilized and/or transformed into more toxic forms (e.g., chromium (III) to chromium (VI)) due to changes in pH and oxidation-reduction potential.
- The compatibility of equipment with the characteristics of the water to which it will be exposed must be considered. Scaling of equipment can be especially problematic when strong bases are introduced into the aquifer and/or highly basic water is extracted from the aquifer. Conversely, corrosion of pipes and equipment may occur when exposed to low pH water.
- It may be more difficult to achieve uniform distribution and target pH throughout the aquifer when introducing strong acids and bases compared to injecting a gas such as CO2. Pockets of highly acidic or highly basic water could persist, which can affect the solubilization of contaminants and naturally occurring minerals, causing sharp increases in groundwater concentrations and potential exceedances of applicable regulatory criteria.
- Formation of scale in the aquifer, wells, and associated piping in systems designed to extract high pH groundwater may result in reduced groundwater injection and/or extraction rates over time. Periodic rehabilitation of the wells and piping may be required to maintain design flowrates.
- Special care should be taken when introducing and mixing strong acids or bases into the aquifer.
- Groundwater mounding may occur during CO2 sparging and should be monitored.
- A vapor recovery system may be required for CO2 applications in and around buildings where the potential for vapor intrusion may be a concern.
- Reapplication of buffers may be required to maintain the pH during bioremediation projects. The frequency of application may be different than that of other amendments (e.g., electron donors).
- The buffering capacity of the aquifer should be determined prior to designing a system to address pH changes. Buffering capacity can be determined using several types of acid-base titrations. If buffering will be performed to counter effects of a remediation system, a titration method that does not include effects of silica dissolution should be used, since silica dissolution is a slow process and most remediation systems require short equilibration times (SERDP and ESTCP, 2019).
- Aquifer pH can be low because of high concentrations of dissolved CO2 in groundwater. When a sample is collected at the surface, the reduced pressure results in CO2 degassing, which will increase the pH of the sample, and therefore the measured acidity will not be representative of the "in situ" conditions at depth.
- The transmissivity of the aquifer could be impacted by changes in pH due to the formation of precipitates that could block flow pathways.
- Special care must be taken when handling strong acids and bases to ensure safety of workers and the surrounding community.
- A portion of injected CO2 will vent to the atmosphere. However, a large fraction may dissolve and remain in the aquifer after injection activities have ceased and may provide long-term neutralization and mitigate potential for rebound.
- Injection of high-pH solutions in sediments can cause clay minerals to disperse and clog the formation near the well. At pH values greater than 10, dissolution and /reprecipitation of silicate minerals may clog the formation (ESTCP and SERDP, 2019).
Resources
American Public Health Association, American Water Works Association, Water Environment Federation. Standard Method 2310 Acidity (2017)
This analytical method describes methods to measure the pH of water and how strong mineral acids, weak acids, and hydrolyzing salts impact the results.
Buddhima Indraratna, Gyanendra Regmi, Long Nghiem, A. Golab. Performance of a PRB for the Remediation of Acidic Groundwater in Acid Sulfate Soil Terrain (2010) (34 pp, 0.5 MB)
Journal of Geotechnical and Geoenvironmental Engineering, 136(7), 2010, 897-906
This journal article describes the results of a demonstration to install and operate a permeable reactive barrier constructed with recycled concrete to treat acidic groundwater resulting from pyrite oxidation of acid sulfate soils.
Groundwater Remediation Technologies Analysis Center. In Situ Flushing (1997) (PDF) (24 pp, 0.2 MB)
This report provides an overview of the general principals and techniques, applicability, and limitations of soil flushing.
Mutch Associates (Mutch). CO2 Sparging Phase 3 Full-Scale Implementation and Monitoring Report (2016) (PDF) (469 pp, 33.9 MB)
This report describes the implementation and monitoring results related to a project to perform CO2 sparging in groundwater to reduce the pH of an alkaline aquifer.
Robinson, Clare, Barry, D.A. Design tool for Estimation of Buffer Requirement for Enhanced Reductive Dechlorination of Chlorinated Solvents in Groundwater (7 pp, 356 KB)
This paper describes the application of BUCHLORIC to calculate the amount of buffer required to maintain the groundwater pH in a DNAPL treatment zone within the optimal range for dechlorinating bacteria.
SERDP and ESTCP. pH Buffering in Aquifers
This website provides an overview of buffering processes to mitigate pH changes in an aquifer during remedial actions.
USEPA. Chemical Enhancements to Pump and Treat Remediation (1992) (20 pp, 370 KB)
This paper describes impacts of pH on groundwater pump and treat systems and describes various enhancements to improve treatment and performance.
