Solidification and Stabilization
On this page:
- Schematic
- Introduction
- Other Technology Names
- Description
- Development Status
- Applicability
- Cost
- Duration
- Implementability Considerations
- Resources
Schematic
This information may be reproduced without restriction as long as the source attribution is included.
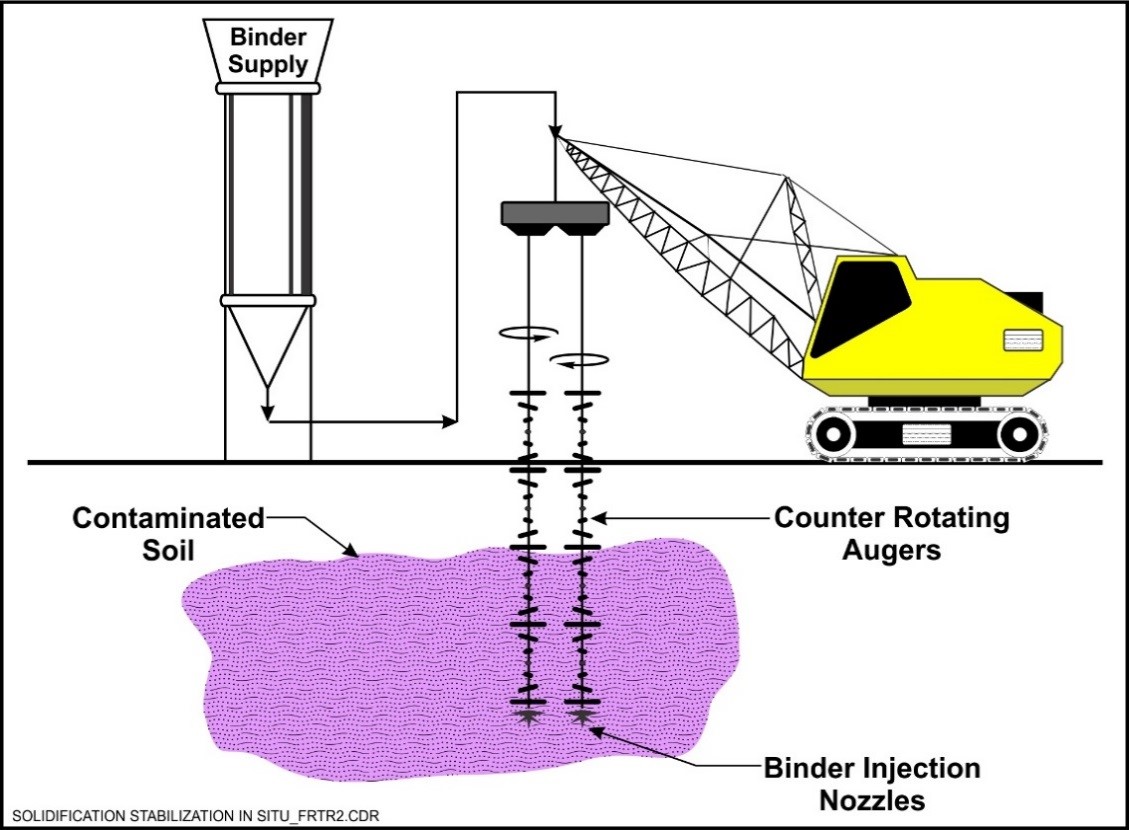
In Situ Solidification/Stabilization
This information may be reproduced without restriction as long as the source attribution is included.
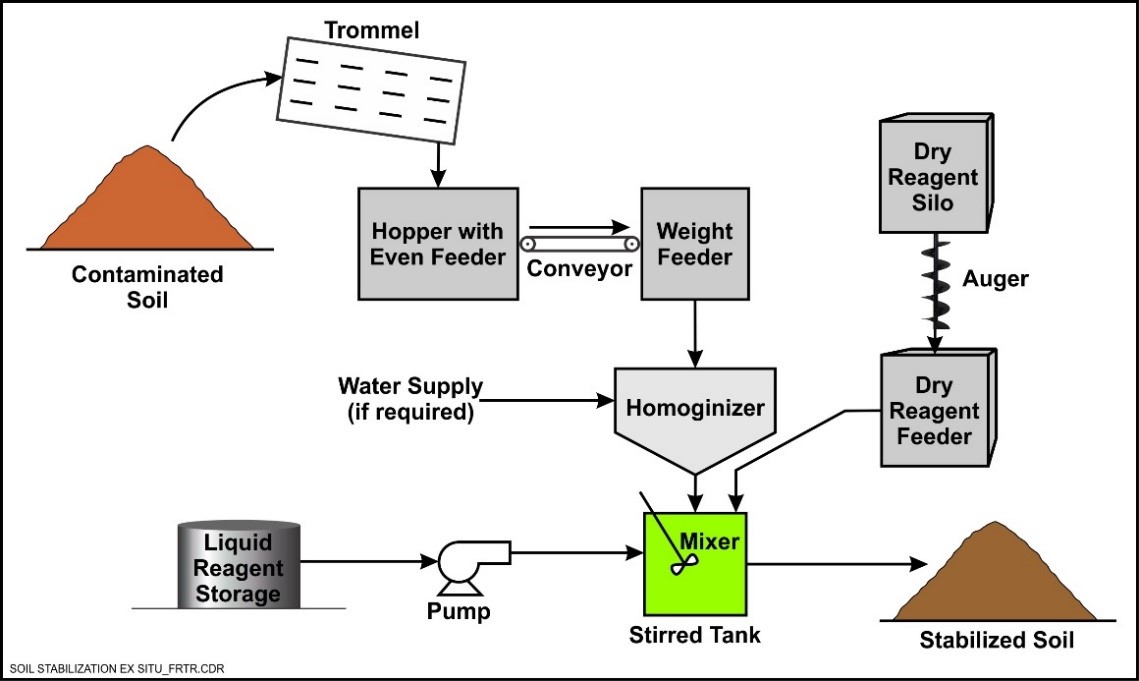
Ex Situ Solidification/Stabilization
Introduction
Solidification/stabilization (S/S) transforms potentially hazardous liquid or solid contaminants of concern (COCs) present in soil or sediment into environmentally innocuous materials of considerably reduced mobility, thus preventing the hazardous waste from reaching receptors. S/S is conducted by mixing various types of binders, additives, and chemicals into the contaminated media to either physically entrap the contaminants (e.g., encapsulation or porosity reduction), or to transform contaminant bonding properties through chemical reaction processes (e.g., hydroxide precipitation), resulting in the formation of highly insoluble salts and hydroxides (e.g., lead phosphate, manganese hydroxide). Mixing can be performed either in situ or ex situ.
Other Technology Names
Encapsulation (e.g., A landfill with sealing liners in all six directions [north, south, east, west, up, and down])
Fixation (i.e., Contaminated media mixed with portland cement, or an agent added to create an insoluble salt of the contaminant (e.g., trisodium phosphate added to lead-contaminated soil to form lead phosphate)
Vitrification (i.e., contaminated solid media heated to its melting point to fix the contamination in a glassy substance)
Bituminization (i.e., encapsulating in tar or asphalt to prevent migration)
Description
S/S treatment is designed to immobilize contaminants within the media (soil or sediment) matrix, rather than removing them through chemical or physical treatment. Solidification refers to a process that uses a reagent to bind the contaminated media to encapsulate the COCs. Solidification adds a binder to the media in order to change the physical properties of the media, which typically results in a decrease in its permeability and an increase in its compressive strength. Stabilization involves a chemical reaction that reduces the leachability of the COCs by chemically immobilizing them and/or reduces their solubility, which results in a reduction in mobility and hence toxicity.
In situ S/S generally 1 requires the addition of a binding agent. Typically, the binding agent is selected based upon the contaminant being treated as well as the soil/sediment medium being processed. Binder materials include inorganic binders (e.g., portland cement, fly ash, lime, soluble silicates and sulfur-based binders), and organic binders (e.g., asphalt, epoxide, granulated activated carbon, organophilic clays, polyesters, and polyethylene). In many cases, the organic binders are applied to immobilize organic COCs prior to adding an inorganic binder to solidify the media.
Inorganic binders are generally less expensive and easier to apply than organic binders. Organic binders are usually applied to the stabilization of radioactive wastes or organic compounds (e.g., polychlorinated biphenyls [PCBs] and polycyclic aromatic hydrocarbons [PAHs]). Selection and appropriate concentration of a suitable binder depend on site-specific factors including the COCs to be treated, the concentration of the COCs in the contaminated soil/sediment media, geophysical properties of the media, and project/site remedial goals and performance criteria, such as desired unconsolidated compressive strength or leachability requirements. In general, the volume of reagent required for in situ treatment can range from 5 to 30 percent per volume of media treated. Bench- and field-scale treatability studies should be employed to determine the optimum binder(s) and appropriate dosage concentrations.
S/S can be applied in situ or ex situ. In situ applications utilize several different methods to mix the binding reagents into the contaminated media. Auger mixing involves using large soil augers to mix the binder directly into soil, which is introduced through nozzles at the bottom of the augers as they turn. Another method is grout injection, which uses high pressure to force the binder into the soil pore space through grout injection pipes. Both techniques can be used to treat media as deep as 60 ft below ground surface. Although deeper treatment can be performed, the cost can become prohibitive.
Shallow contamination can be stabilized using common excavation equipment. A very rudimentary technique is to use an excavator and bucket to mix in the reagents at the surface. Success of this technique is dependent on the experience of the operator and the amount of time that is spent mixing the materials within each treatment cell. More specialized types of equipment also can be used including excavators equipped with forks configured in a rake-like fashion, through which the amendments are injected and high-speed rotary blenders that utilize a cutting head designed to facilitate mixing of the amendments and contaminated media. Shallow mixing techniques generally are applicable to a maximum depth of about 10 feet. They are not very effective at sites where a substantial amount of debris is present.
Ex situ S/S is performed by excavating the soil or sediment, separating out debris, and then mixing the soil with stabilizing agents in either a rotary drum or pug mill. Ex situ S/S can be performed on or off site. In either case, the resulting materials must be disposed. However, if treatment is performed at the site, the treated material may be able to be placed back into the excavation, without triggering land disposal restrictions if a Corrective Action Management Unit (CAMU) designation is obtained. The primary advantage of ex situ S/S is that more uniform mixing can generally be achieved than in situ applications; however, the increased cost to excavate and dispose of the material must be considered.
There are many variations of S/S technology that can be applied to treat soils and sediments contaminated with various COCs and create various end products that must be disposed or recycled. These variations fall into four categories based on the binders used, including (Raj et al., 2005):
- Inorganic cementation systems that incorporate portland cement, lime, and other pozzolans.
- Organic binders including asphalt, polyethylene emulsions, bitumen, and other thermoplastics.
- Thermosetting organic polymers including epoxides, phenolics, and urea.
- Vitrification, which relies on high temperature and mixing the waste with glass-forming substances to jointly melt them and then solidify the waste material as it cools. Due to its high cost, it generally is reserved for treatment of radioactive/hazardous waste mixtures.
Common S/S processes included within these categories are:
- Bituminization, which is an ex situ process that embeds wastes in molten bitumen and becomes encapsulated when the bitumen cools. The process combines heated bitumen and a concentrate of the waste material, usually in slurry form, in a heated extruder containing screws that mix the bitumen and waste. Water is evaporated from the mixture to about 0.5% moisture. The final product is a homogenous mixture of extruded solids and bitumen. This process can be used to encapsulate radioactive waste and greatly reducing alpha and beta radiation emitted from it.
- Emulsified asphalt is an ex situ process used to stabilize soils contaminated with petroleum hydrocarbons and other organic contaminants (Conway, 1993). Asphalt emulsions, which can be cationic or anionic, are very fine droplets of asphalt dispersed in water that are stabilized by chemical emulsifying agents. These emulsions are mixed into the soil, after which the emulsion breaks and the water in the waste is released. The organic phase forms a continuous matrix of hydrophobic asphalt around the waste solids, which is uniformly distributed throughout the soil. After sufficient time to set and cure, the resulting solid asphalt is impermeable to water. Water released from the waste is treated and/or disposed in accordance with local regulations.
- Modified sulfur cement is a commercially-available thermoplastic material, a substance that becomes plastic on heating and hardens upon cooling, which can be used to stabilize soil and sediment contaminated with radionuclides and heavy metals. It is easily melted (127° to 149° C [260° to 300° F]) and then mixed ex situ with the waste to form a homogenous molten slurry which is discharged to suitable containers for cooling, storage, and disposal. The relatively low temperatures used limit emissions of sulfur dioxide and hydrogen sulfide to allowable threshold values.
- Polyethylene extrusion is an ex situ process that involves the mixing of polyethylene binders and dry waste materials using a heated cylinder containing a mixing/transport screw. The heated, homogenous mixture exits the cylinder through an output die into a mold, where it cools and solidifies. The properties of the polyethylene produce a very stable, solidified product. The process has been tested on nitrate salt wastes at pilot scale, thereby establishing its viability, and has been demonstrated to treat radioactive and mixed hazardous waste (Lageraaen and Kalb, 1997).
- Pozzolan/portland cement stabilization, which can be performed in situ or ex situ, primarily consists of mixing silicates from pozzolanic-based materials like fly ash, kiln dust, pumice, or blast furnace slag and cement-based materials like portland cement. These materials chemically react with water to form a solid cementitious matrix that improves the handling and physical characteristics of the waste. They also raise the pH of the water, which may help precipitate and immobilize some heavy metal contaminants. Pozzolanic and cement-based binding agents are appropriate for inorganic contaminants, primarily heavy metals. The effectiveness of this binding agent with organic contaminants varies.
- Sludge stabilization, which is performed ex situ, includes dewatering as needed followed by the addition of a reagent, either slags or cementitious materials, to a sludge to transform the material so that the hazardous constituents are in their least mobile or toxic form. Sludges that leach heavy metals or other contaminants are often stabilized to immobilize the hazardous constituents.
- Soluble phosphate stabilization involves the addition of various forms of phosphate and alkali for control of pH as well as for formation of complex metal molecules of low solubility to immobilize (insolubilize) metals over a wide pH range. It can be performed in situ or ex situ. It has been primarily used to stabilize lead and other heavy metals contamination. Unlike most other stabilization processes, soluble phosphate processes do not convert the waste into a hardened, monolithic mass.
- Vitrification is a related S/S technology (performed either ex situ or in situ) that employs a different mechanism to contain contaminants. It uses an electric current to melt soil or other earthen materials at extremely high temperatures (up to 1,200°C) to melt and convert waste materials into glass or other glass and crystalline products. The high temperatures destroy any organic constituents with very few byproducts. Contaminants, such as heavy metals and radionuclides, are incorporated into the glass structure, which is generally a relatively strong, durable material that is resistant to leaching. In addition to solids, the waste materials can be liquids, wet or dry sludges, or combustible materials. Borosilicate and soda lime are the principal glass formers and provide the basic matrix of the vitrified product. Vitrification can be used to treat a wide range of contaminants. It completely oxidizes organic contaminants such as volatile organic compounds (VOCs), semi-organic compounds (SVOCs), dioxins, and PCBs, and will immobilize metals and radionuclides within the resulting solid matrix.
Effectiveness of in situ S/S relies on successful implementation during the construction phase. The formulations developed based on treatability tests must be achieved in the field. It is important to ensure that the correct proportions are attained, and that adequate mixing is imparted to the material. Specifically, treatability testing should at a minimum:
- Determine the most appropriate binding agents.
- Determine the impact of the binding agents on all contaminants and other materials present in the media.
- Assess if emissions of contaminants during implementation will occur and require mitigation.
- Assess the volume increase that may occur as a result of adding the binding agents.
Parameters that affect the blending of contaminated materials, such as mixing speed, reagent, delivery rate, soil treatment rate, and mixing time, must be carefully monitored during application. In addition, performance criteria must be established prior to implementation. Typically, these criteria will be established for strength (typically unconfined compressive strength), hydraulic conductivity, and leachability. Specific criteria and values are based on remedial goals for the site as well as future anticipated land use. Samples are collected during implementation and tested. Results are compared to the performance criteria to gauge the degree to which the remedy is performing and if any modification to the formula is necessary.
Development Status and Availability
Development and Implementation Status The following checklist provides a summary of the development and implementation status of S/S technologies:
☐ At the laboratory/bench scale and shows promise
☐ In pilot studies
☒ At full scale
☐ To remediate an entire site (source in vadose zone)
☒ To remediate a source only
☒ As part of a technology train
☐ As the final remedy at multiple sites
☐ To successfully attain cleanup goals in multiple sites
S/S technologies are available:
☒ Commercially available nationwide
☐ Commercially available through limited vendors because of licensing or specialized equipment
☐ Research organizations and academia
Applicability
|
Contaminant Class Applicability Rating for Solidification and Stabilization (Rating codes: Demonstrated Effectiveness, ◐ Limited Effectiveness, No Demonstrated Effectiveness, I/D Insufficient Data, N/A Not Applicable) |
||||||||
|---|---|---|---|---|---|---|---|---|
Nonhalogenated VOC |
Halogenated VOC |
Nonhalogenated SVOC |
Halogenated SVOC |
Fuels |
Inorganics |
Radionuclides |
Munitions |
Emerging Contaminants |
| ○ | ○ | ◐ | ◐ | ◐ | ● | ● | ● | N/A |
S/S technologies are applicable to a broad range of contaminants because of the many variations of the technology and reagents that can be applied. Application of this technology has been successfully performed to treat metals and radioactive materials. Improved binder formulations have led to successful treatment efficiency of organic COCs including non-volatile compounds and SVOCs such as chlorinated ethenes, polycyclic aromatic hydrocarbons, PCBs, pesticides, and dioxans and furans. However, some organic compounds at certain concentrations can have a detrimental effect on the properties of cementitious materials; hence, they may interfere with the binding process. With the exception of vitrification, S/S does not effectively treat VOCs. As a result, VOCs may not be immobilized and leachability and strength performance requirements may not be achieved. The use of certain materials, such as organophilic clay and activated carbon, can improve contaminant immobilization in the solidified/stabilized wastes. S/S is expected to treat emerging contaminants that bond to soil, however, its effectiveness to treat compounds that have high aqueous solubilities and little affinity for soil such as 1,4-dioxane and perfluoroalkyl substances (PFAS) may be limited, and additional research is needed. Recently, ex situ stabilization of PFAS using a mixture of activated carbon and other stabilizing materials has shown promise for stabilizing PFAS (Sörengård et al., 2019).
Cost
The cost of S/S technologies is dependent on the type of treatment applied and whether an in situ or ex situ process is employed. Costs vary widely according to materials or reagents used, their availability, project size, and chemical nature of contaminants. Major cost drivers include:
Upfront Costs
- In situ or ex situ application. Additional costs are associated with ex situ applications due to the need to excavate and place it back into the excavation and/or dispose of the stabilized material. However, treatment generally is faster.
- Area and depth of contaminants requiring treatment, which impact type and size of equipment and time required on site, and excavation costs in the case of ex situ applications.
- Moisture content, which impacts the volume of reagent that must be added. As the moisture content increases, the amount of reagent required for adequate treatment also increases.
- Nature or extent of contamination, which impacts the type of S/S process applied and equipment required as well as the quantities and nature of the reagents that must be added.
- Complexity of S/S equipment.
Operation and Maintenance Costs
- Types and quantities of reagents needed.
- Resulting volume of the treated media; the addition of the reagents can result in substantial increase in the volume of media treated. Hence, additional costs associated with removal/disposal or other infrastructure needs to accommodate the additional material may be incurred.
- Dewatering costs, which may be required for in situ application at sites where the contaminated media is located in the saturated zone. Dewatering and treatment can be a substantial cost.
- Transportation costs for off-site disposal of material generated during ex situ applications.
- Long-term monitoring for in situ applications.
The list above highlights those cost dependencies specific to S/S technologies and does not consider the dependencies that are general to most in situ remediation technologies. Click here for a general discussion on costing which includes definitions and repetitive costs for remediation technologies. A project-specific cost estimate can be obtained using an integrated cost-estimating application such as RACER® or consulting with a subject matter expert.
Duration
S/S is a short- to medium-term treatment technology expected to require weeks to months to achieve treatment objectives. Ex situ processes are limited by the time it takes to excavate the soil as well as the production rate of the treatment plant. Production rates will vary depending on the type of processes used, required mixing times, and time needed for the desired reactions to occur. The durations for in situ applications also are dependent on numerous site-specific factors, including:
- Depth and volume of contamination requiring treatment
- Contaminant distribution
- Mixing method
- Subsurface heterogeneity, soil characteristics including particle size distribution and density
- Quantity of reliable mixing equipment available on site.
- Diameter of auger (in case of auger mixing)
- Presence of subsurface utilities or debris
- Weather conditions
The Environmental Protection Agency (EPA) analyzed data on completed S/S projects at Superfund sites to determine the average operational time for S/S projects compared to other technologies. Based on its findings, assuming treatment of 1,000 cubic yards of waste, the average operational time was 1.1 months (EPA, 2000).
Implementability Considerations
The following are key considerations associated with applying S/S technologies:
- Depth of contaminants limit the potential for ex situ applications and may limit some types of in situ application processes, such as soil mixing. For example, auger mixing is limited to depths less than 100 ft. However, site benching (creating steps in the sides of an excavation) and sloping (forming sides of an excavation inclined away from the excavation bottom) can be performed to increase the maximum depth of an excavation.
- Ex situ applications can achieve better mixing and homogeneous properties in the stabilized material than in situ applications due to inherent heterogeneities in the subsurface.
- For in situ applications, the solidified material may prevent certain future site uses. Also, future site use may "weather" the materials and affect the ability to maintain immobilization of contaminants. Long-term monitoring and institutional controls may be required for an indefinite period of time. Bench-scale testing for in situ applications should include long-term stability testing over relevant weather/climate cycles, simulation of infiltration/groundwater flow, etc.
- S/S generally is an irreversible process if designed and applied correctly taking into consideration soil and groundwater chemistry and the types and concentrations of contaminants present. Because of the wide range of S/S techniques available, types of binding agents that can be used, and types of contaminants/wastes that can be treated it is important to perform bench and/or pilot tests to design a remedy that will ensure the contamination remains stabilized for the length of time required based on disposal or end use requirements.
- S/S techniques are not designed to decrease contaminant toxicity, but to control migration and contact with receptors.
- Some processes result in a significant increase in volume (up to double the original volume), which must be disposed. For in situ applications, this increase in the treated volume must be accounted for by either grading or removal of additional soil volume.
- Certain wastes are incompatible with variations of this process. Treatability studies are generally required.
- Reagent delivery and effective mixing for in situ applications are more difficult than for ex situ applications.
- Like all in situ treatments, confirmatory sampling can be more difficult than for ex situ treatments.
- For in situ applications, processing of contaminated soil below the water table may require dewatering.
- Rocks and debris must be removed for ex situ applications and can interfere with the adequate introduction and mixing of the binding reagents during in situ applications.
- For in situ applications, binder injection and mixing must be controlled to minimize the spread of contaminants to clean areas.
- For in situ treatment, influences of the treated material monolith on local groundwater flow conditions must be evaluated, as well as the effects of infiltration and groundwater contact on long-term stability.
- Although complex waste mixtures can be treated, it can be difficult to formulate an effective binder for heterogeneous mixtures.
- S/S can improve structural properties of soil, waste, and sludge (e.g., strength) to facilitate consideration of land beneficial reuse.
- On-site management of contaminated materials conserves landfill space with no transportation off site.
- Impacts of changing groundwater elevation on the stabilized waste material should be evaluated for in situ applications and at disposal locations for ex situ applications.
- Injecting grout or other stabilization materials at high pressure can cause fracturing, which could spread contamination if not properly designed and applied, and therefore might raise regulatory and stakeholder concern. In addition, because it may not be possible to inject and mix the grout uniformly, it is difficult to achieve a homogenous, stabilized mass and preferential pathways through the stabilized mass can result.
- Vapor treatment may be required for some S/S projects. For example, if reagents, such as quicklime, which quickly produce heat are used, waste-containing VOCs may generate high concentrations of VOCs in air during treatment. Also, if mercury is present in waste treated by vitrification, vapor treatment equipment will be required to capture mercury that is volatilized.
Resources
Brookhaven National Laboratory (BNL). Use of Recycled Polymers for Encapsulation of Radioactive, Hazardous and Mixed Wastes (1997)
(PDF) (28 pp, 457 KB)
This report describes polymer encapsulation technology.
Conway, Michael F. Bench-Scale Evaluation of Asphalt Emulsion Stabilization of Contaminated Soils (1993), Journal of Soil Contamination
This journal article describes the results of a bench-scale test to evaluate the effectiveness of using an asphalt emulsion to immobilize contaminants from petroleum products.
DOE. Hanford Vitrification Plant
This website describes vitrification and provides information about the Hanford Vitrification Plant that was constructed to convert liquid radioactive and chemical waste into a solid glass.
DOE. In Situ Vitrification Applied to Buried Wastes (1992)
(PDF) (84 pp, 5.16 MB)
This technology status bulletin describes the in situ vitrification process and its application to the subsurface.
EPA. Solidification/Stabilization of Organics and Inorganics (1993)
(PDF) (13 pp, 1.70 MB)
This engineering bulletin describes S/S and in situ and ex situ applications to treat inorganics, radionuclides, and some organic wastes.
EPA. Stabilization/Solidification Processes for Mixed Waste (1996)
(PDF) (91 pp, 971 KB)
This report describes grout/portland cement stabilization, sulfur polymer encapsulation, polymer encapsulation and Phoenix ash technology S/S processes.
EPA. Solidification/Stabilization Resource Guide (1999)
(PDF) (91 pp, 960 KB)
This resource guide provides information on published materials such as field reports and guidance documents that address issues relevant to S/S technologies.
EPA. Solidification/Stabilization Use at Superfund Sites (2000)
(PDF) (23 pp, 988 KB)
This document provides statistics pertaining to the application of S/S at Superfund sites.
EPA. Biosolids Technology Fact Sheet (2000)
(PDF) (9 pp, 185 KB)
This fact sheet describes the use of alkaline stabilization to treat biosolids.
EPA. Soil Washing Treatment (1990)
This engineering bulletin describes the soil washing process and its applicability to treat a wide range of contaminants.
EPA. A Citizens Guide to Solidification/Stabilization (2001)
(PDF) (2 pp, 774 KB)
A fact sheet intended for public guidance on the method of S/S to clean up pollution at Superfund and other sites.
EPA. Technology Performance Review: Selecting and Using Solidification/Stabilization Treatment for Site Remediation (2009)
(PDF) (28 pp, 863 KB)
This summary report provides reviews of a variety of S/S treatments.
FRTR. Remediation Case Studies: Thermal Desorption, Soil Washing, and In Situ Vitrification (1995)
(PDF) (174 pp, 8.23 MB)
This report provides several case studies of site cleanups using thermal desorption, soil washing and in situ vitrification.
Hensley, Timothy T. Pozzolan Stabilized Subgrades Nebraska Department of Roads Research Project (2007)
(PDF) (51 pp, 1.52 MB)
This report describes results of research to investigate the performance of lime, cement kiln dust and fly ash for use as stabilization agents with a variety of Nebraska soils.
ITRC. In Situ Stabilization/In-place Inactivation (1997)
This document describes in situ stabilization/in-place inactivation as a technology for the remediation of metals in soil. It outlines several case studies and identifies future research and development needs as of 1997.
ITRC. Technology Overview Chemical Stabilization Phosphate and Biosolids Treatment (2010)
(PDF) (17 pp, 230 KB)
This report describes the application of phosphate and biosolids to treat soils contaminated with heavy metals.
ITRC. Development of Performance Specifications for Solidification/Stabilization (2011)
This document provides guidance on comprehensive performance specifications, applicable contaminants, effectiveness, and long-term performance of S/S.
P.D.Kalb, J.H.Heiser III, P.Colombo. Modified Sulfur Cement Encapsulation of Mixed Waste Contaminated Incinerator Fly Ash. Journal of Waste Management. Volume 11, Issue 3. 1991
This journal article describes a process using modified sulfur cement, a thermoplastic material to stabilize mixed waste contaminated incinerator fly ash.
Lin, Sheng-Lung, Lai, James, S., and Chian, Edward, S. K. Modifications of Sulfur Polymer Cement (SPC) Stabilization and Solidification (S/S) Process (1995), Waste Management, Vol. 15
This paper addresses the effectiveness of using sulfur polymer cement to stabilize lead-contaminated soils.
Mattias Sörengård, Dan B. Klejab, Lutz Ahrensa. Stabilization and Solidification Remediation of Soil Contaminated with Poly and Perfluoroalkyl Substances (PFASs). Journal of Hazardous Materials. Volume 367, pages 639-646 (2019)
This journal article describes the results of bench-scale tests to evaluate the efficacy of S/S to treat soil contaminated with PFAS.
In situ vitrification requires heat as opposed to a binding agent. In addition, some stabilization reactions, such as mercury stabilization with sulfur, impact the speciation of the metal, thereby decreasing bioavailability and mobility, without the need for a binder. ↩
In situ vitrification requires heat as opposed to a binding agent. In addition, some stabilization reactions, such as mercury stabilization with sulfur, impact the speciation of the metal, thereby decreasing bioavailability and mobility, without the need for a binder. ↩
